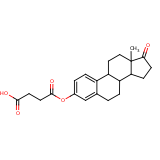
| Compound Information | SONAR Target prediction | | Name: | ESTRONE HEMISUCCINATE | | Unique Identifier: | SPE01503676 | | MolClass: | Checkout models in ver1.5 and ver1.0 | | Molecular Formula: | C22H26O5 | | Molecular Weight: | 344.232 g/mol | | X log p: | 5.334 (online calculus) | | Lipinksi Failures | 1 | | TPSA | 60.44 | | Hydrogen Bond Donor Count: | 0 | | Hydrogen Bond Acceptors Count: | 5 | | Rotatable Bond Count: | 5 | | Canonical Smiles: | CC12CCC3C(CCc4cc(OC(=O)CCC(O)=O)ccc34)C1CCC2=O | | Therapeutics: | estrogen |
| Species: |
4932 |
| Condition: |
ARF1 |
| Replicates: |
2 |
| Raw OD Value: r im |
0.6225±0.0127279 |
| Normalized OD Score: sc h |
0.9159±0.00931876 |
| Z-Score: |
-4.0765±0.464863 |
| p-Value: |
0.0000944774 |
| Z-Factor: |
-21.344 |
| Fitness Defect: |
9.2671 |
| Bioactivity Statement: |
Active |
| Experimental Conditions | | | Library: | Spectrum | | Plate Number and Position: | 22|H9 | | Drug Concentration: | 50.00 nM | | OD Absorbance: | 600 nm | | Robot Temperature: | 26.10 Celcius | | Date: | 2007-10-02 YYYY-MM-DD | | Plate CH Control (+): | 0.041175±0.00062 | | Plate DMSO Control (-): | 0.6629750000000001±0.22109 | | Plate Z-Factor: | -0.2493 |
|  png png
ps
pdf |
| 3274 |
4-[(13-methyl-17-oxo-7,8,9,11,12,14,15,16-octahydro-6H-cyclopenta[a]phenanthren-3-yl)oxy]-4-oxo-butanoic
acid |
| 5702254 |
4-[[(13S)-13-methyl-17-oxo-7,8,9,11,12,14,15,16-octahydro-6H-cyclopenta[a]phenanthren-3-yl]oxy]-4-oxo-bu
tanoic acid |
| 6553596 |
4-[[(8R,9R,13S,14R)-13-methyl-17-oxo-7,8,9,11,12,14,15,16-octahydro-6H-cyclopenta[a]phenanthren-3-yl]oxy
]-4-oxo-butanoic acid |
| 6978338 |
4-[[(8R,9R,13R,14R)-13-methyl-17-oxo-7,8,9,11,12,14,15,16-octahydro-6H-cyclopenta[a]phenanthren-3-yl]oxy
]-4-oxo-butanoate |
| 6978339 |
4-[[(8R,9R,13R,14R)-13-methyl-17-oxo-7,8,9,11,12,14,15,16-octahydro-6H-cyclopenta[a]phenanthren-3-yl]oxy
]-4-oxo-butanoic acid |
| 7067988 |
4-[[(8R,9S,13S,14R)-13-methyl-17-oxo-7,8,9,11,12,14,15,16-octahydro-6H-cyclopenta[a]phenanthren-3-yl]oxy
]-4-oxo-butanoate |
| internal high similarity DBLink | Rows returned: 0 | |
| active | Cluster 13348 | Additional Members: 15 | Rows returned: 2 | |
|
